I don’t know about you, but I hate chemistry. I battled through every one of my classes and failed two of them. I played dirty, and so did the chemistry. I cried a lot. But in the end, I’m grateful to know the damn stuff. We know the minute structure of the building blocks of life, matter, all things.
These new images should get shown in every chemistry class from now on:
Y’all, I’m tempted to say that this is the coolest thing I’ve ever seen. I do tend to be too superlative, but wow. These photos are nuts.
Atoms of each chemical element have their own unique number of electrons, and depending on that number, different elements interact with each other in wildly variable, but individually predictable, ways. So when we throw atoms together to make larger aggregations of matter, they tend to bond to each other in predictable molecular structures, based on the numbers of electrons they all bring to the group.
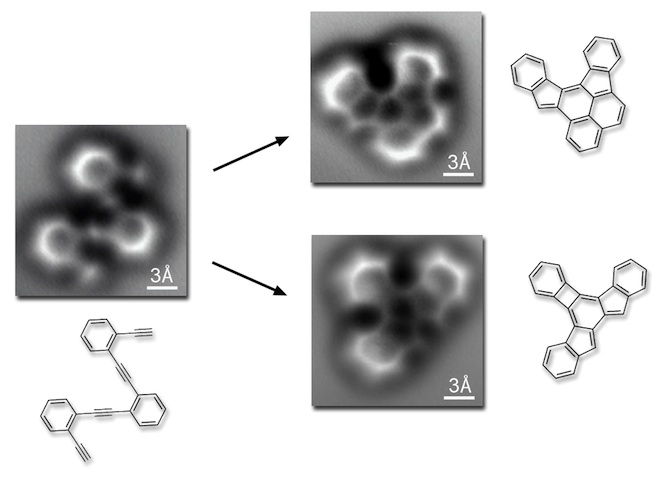
This is the FIRST TIME anyone has gotten images of the bonds that the atoms form between each other. These are the structures I drew out, torturously, repetitively, again and again in my lab notebooks til I thought my hands would fall off—these are the structures of molecules, of matter. We’ve never seen them before! And just look how right we are about those structures, how visible that geometry actually is! You can even see, in those brighter spots, places where the bonds are doubled and stronger—represented by a double line in the drawings. This is just unreal, folks.
The research was published in this paper in May, and written up very nicely in this article from WIRED.
Carbons eating their own tails
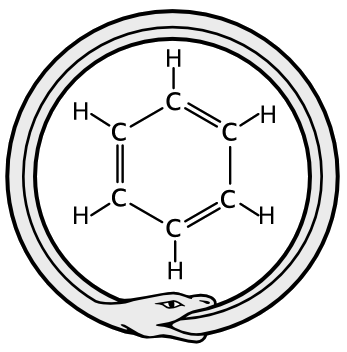
Now for my favorite chemistry story. You see those six-sided rings with a lot of double bonds? Those are benzene rings: neat rings of six carbon atoms that are very stable. It’s very important in organic functions, partially for its stability: carbon is one of the main building blocks of living matter.
For a long time, chemists knew that carbon liked to team up in stable groups of six. But when they tried to draw the molecule, with bonds—one carbon over here, holding hands with this next one, and the next—they found it impossible to draw something that made sense, given the number of electrons carbon atoms have. The scientists were stumped for years.
One man, August Kekulé, ruminated and struggled over benzene with all the rest. Then, as the story goes, he had a dream, or a day-dream, or a vision. He saw a mythical Ouroboros, a snake curled up, eating its own tail. Suddenly he knew that the carbon atoms were joined in a ring, and with a rightly satisfying alternating pattern of single and double bonds, the structure worked:
The veracity of this delightful story is debated, as the greatest stories always are. But luckily not too much–we can probably go ahead and believe it, and feel alright about ourselves.
And if it is true, August Kekulé had his dream around 1865. Look back up at those images, taken in 2013. There are your benzene rings, with bonds in place right where they should be. I can only imagine that if Kekulé could see them, he would be overjoyed.
Maybe I don’t hate chemistry after all.
you are a science geek!
logo
dr deborah bernstein warwick, new york (845) 986-6684
http://www.drdeb.biz
logo